Tartrazine alaminium lake
Food Yellow 4:1
CAS: 12225-21-7
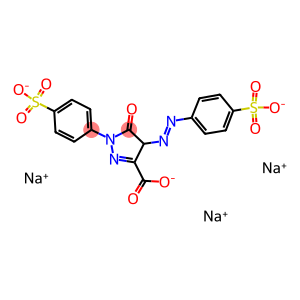
Molecular Formula: C16H9N4O9S2.3Na
Tartrazine alaminium lake - Names and Identifiers
| Name | Food Yellow 4:1 |
| Synonyms | KI4 C.I. Pigment Yellow 100 Tartrazine alaminium lake C.I. Acid Yellow 23-aluminum lake 1H-Pyrazole-3-carboxylic acid, 4,5-dihydro-5-oxo-1-(4-sulfophenyl)-4-((4-sulfophenyl)azo)- aluminum salt aluminum tris(4-{(E)-[3-carboxy-5-oxo-1-(4-sulfophenyl)-4,5-dihydro-1H-pyrazol-4-yl]diazenyl}benzenesulfonate) |
| CAS | 12225-21-7 |
| EINECS | 235-428-9 |
| InChI | InChI=1/3C16H12N4O9S2.Al/c3*21-15-13(18-17-9-1-5-11(6-2-9)30(24,25)26)14(16(22)23)19-20(15)10-3-7-12(8-4-10)31(27,28)29;/h3*1-8,13H,(H,22,23)(H,24,25,26)(H,27,28,29);/q;;;+3/p-3/b3*18-17+ |
Tartrazine alaminium lake - Physico-chemical Properties
| Molecular Formula | C16H9N4O9S2.3Na |
| Molar Mass | 534.368 |
| Density | 0.341[at 20℃] |
| Water Solubility | 561.286mg/L at 30℃ |
| Solubility | 600mg/L in organic solvents at 20 ℃ |
| pKa | 0[at 20 ℃] |
| Storage Condition | Room Temprature |
| Physical and Chemical Properties | solubility: slightly soluble in ethanol and Fibrinolysin; Yellow solution in concentrated sulfuric acid, diluted yellow solution, yellow in concentrated nitric acid; Water solution with hydrochloric acid does not change, the concentrated alkali was red and yellow. hue or shade: Green Yellow relative density: 1.78-1.87 Bulk density/(lb/gal):14.8-15.5 oil absorption/(g/100g):33-43 hiding power: transparent reflection curve: 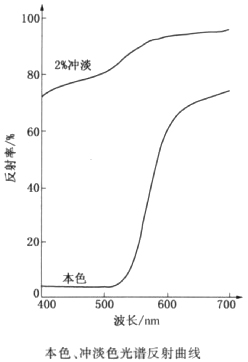 |
| Use | There are 31 types of this product. Given the green light yellow, low coloring strength, alkali and soap resistance performance is poor, only medium baking stability, light fastness is low, little industrial importance. Mainly used in food and medicine and cosmetics coloring, namely food Yellow 5 (FD & D Yellow No 5), occasionally also as a decorative paint, nitrocellulose solvent printing ink and packaging ink and other coloring. |
Tartrazine alaminium lake - Reference
| Reference Show more | 1. [IF=10.588] Chaofan Zheng et al."Modified magnetic chitosan microparticles as novel superior adsorbents with huge “force field” for capturing food dyes."J Hazard Mater. 2019 Apr;367:492 |
Tartrazine alaminium lake - Reference Information
| LogP | 0.028 at 25℃ |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | food yellow 4:1 can be used for fruit juice (flavor) drinks, carbonated drinks, prepared wine, candy, colored cakes, canned watermelon sauce, green plum, lactic acid bacteria beverage, vegetable protein beverage and shrimp (flavor) tablets in 0.10 g/kg; 0.20 g/kg when used for candy coating and red and green silk; for ice cream, the dosage is 0.09 g/kg. |
| identification test | take 0.1g of sample, add 5ml of dilute sulfuric acid test solution (TS-241), mix thoroughly, and fix the volume to 100ml with 0.02mol/L ammonium acetate solution. The solution can be centrifuged if it is opaque. Take l ~ 10ml of this solution to make the measured absorbance within the range of 0.2~0.7, and add 0.02mol/L ammonium acetate solution to 100ml. This solution has a maximum absorption peak at a wavelength of 428nm and 2nm. Take 0.1g of sample, add 5ml of dilute hydrochloric acid test solution (TS-117), and heat it in a water bath for about 5min under constant stirring. After dissolution, it should be almost transparent yellow solution. After cooling, the ammonia test solution (TS-13) should be yellow after neutralization and produce gel-like precipitate of the same color. Take 0.1g of sample, add 5ml of 10% sodium hydroxide solution, shake and heat for about 5min in a water bath, and then almost become transparent and slightly red yellow after dissolution. After cooling, add dilute hydrochloric acid test solution to neutralize, it should be yellow, and produce gel-like precipitate of the same color. Take 0.1g of sample, add 5ml of sulfuric acid, shake and heat for about 5min in a water bath, and it should be yellow. After cooling, take 2~3 drops of the upper clear layer and add them to 5ml of water, which is still yellow. Take 0.1g of sample, add 10ml of dilute hydrochloric acid test solution, and heat it in water bath until most of it is dissolved. Add 0.5g of activated carbon, fully shake, filter, and neutralize the colorless filtrate with 10% sodium hydroxide solution. The aluminum salt test (I-2) should be positive. |
| content analysis | method-titanium trichloride titration method was determined according to the content analysis method in "amaranth red aluminum lake (17005)", but 15g of sodium hydrogen tartrate was used instead of 15g of sodium citrate, 10ml of 0.1% bright green SF was used as indicator, and blank test was carried out at the same time. Accurately weigh the sample in such a way that the consumption of 0.1mol/L titanium trichloride solution is about 20ml. 0.1mol/L titanium trichloride solution per ml is equivalent to 11.71mg of lemon yellow (C16H17N4O9 S2). Method 2 Preparation of standard samples by spectrophotometry 0.500g of lemon yellow aluminum lake with a content of 10.0%, 20ml of water and 2g of sodium hydrogen tartrate, heated to 90 ℃, dissolved, transferred to a 500ml volumetric flask, and fixed volume. Take 10ml of the liquid and then set the volume to 250ml. The sample liquid is prepared according to the standard sample liquid method. The absorbance was measured with a 10mm cuvette at 428nm ± 2nm for the sample and standard sample solution. Water is used as the reference liquid. The ratio of the absorbance of the sample liquid to the standard sample liquid multiplied by the content in the standard sample is the content in the sample. |
| use limit | GB 2760-2000: same as "17070, lemon yellow". |
| use | edible yellow pigment. In Japan, it can be used for grease food, powder food (cake prefabricated powder, powder juice), sugar coating of confectionery products, etc., alone or in combination with other aluminum lakes. The usage of frying oil and shortening is 0.01% ~ 0.02%. When red is enhanced, it is combined with sunset yellow aluminum lake (10% ~ 20%). The coating ability of aluminum lake is much stronger than that of water-soluble food pigment, so it is often used for coloring powder beverages with an amount of 0.01% ~ 0.02%. It can also be used for coloring printing inks, cooking utensils and food containers of food packaging materials. |
| production method | aluminum hydroxide is prepared from aluminum salt such as aluminum sulfate, aluminum chloride and alkali such as sodium carbonate, and lemon yellow aqueous solution is added for precipitation. |
Last Update:2024-04-09 02:00:06
Supplier List
Product Name: Food Yellow 4:1 Request for quotation
CAS: 12225-21-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 12225-21-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Food Yellow 4:1 Visit Supplier Webpage Request for quotation
CAS: 12225-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 12225-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Food Yellow 4:1 Request for quotation
CAS: 12225-21-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 12225-21-7
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Food Yellow 4:1 Visit Supplier Webpage Request for quotation
CAS: 12225-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 12225-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

